- Owl city in christ alone lyrics
- Jio cinema web video not playing
- Real rock riddim my sound champion
- How to install prison architect prisons
- Windows movie maker old version
- Cen-tech 7 function digital multimeter to check car battery
- Robotstudio slamtec download
- Video lucu cak lontong
- Sony xperia st26i unlock setool
- Crazy taxi 3 pc patch
- 2020 hummer h3
- East west symphonic choirs modulation not working
- Mtp stock
- Autocad drawing online
- Gps pathfinder office 5-3 crack
- Harley quinn and poison ivy

Standard & Poor's and S&P are registered trademarks of Standard & Poor's Financial Services LLC and Dow Jones is a registered trademark of Dow Jones Trademark Holdings LLC. Dow Jones: The Dow Jones branded indices are proprietary to and are calculated, distributed and marketed by DJI Opco, a subsidiary of S&P Dow Jones Indices LLC and have been licensed for use to S&P Opco, LLC and CNN.
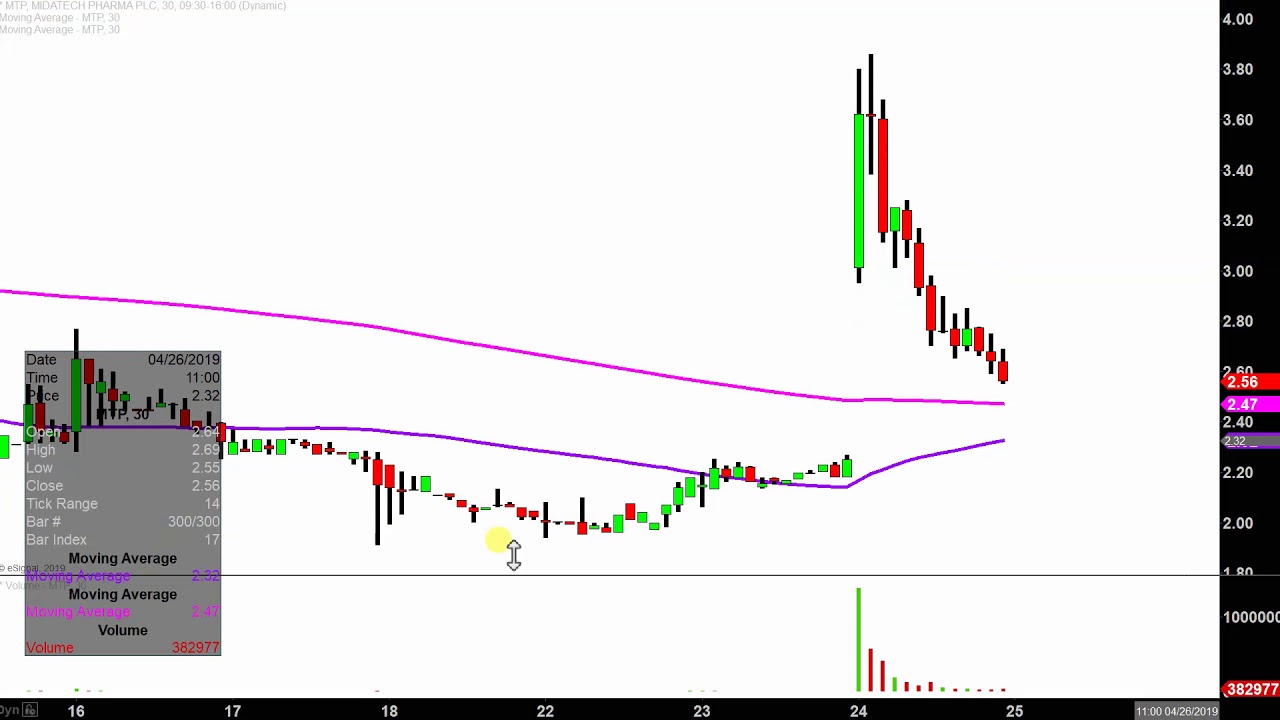
#MTP STOCK PATCH#
The polarity and gender of your patch cords will depend on your network layout and type of MTP (MPO) adaptor used. Chicago Mercantile Association: Certain market data is the property of Chicago Mercantile Exchange Inc. Patch to MTP (MPO) trunk cables We also stock MTP (MPO) patch cables for the second application and these will typically be polarity B but will have different gender connectors (Male to Female). Factset: FactSet Research Systems Inc.2019. Market indices are shown in real time, except for the DJIA, which is delayed by two minutes. Before making any investment, you should do your own analysis.Most stock quote data provided by BATS. But there can be no assurance on the timing for concluding the discussions nor any assurance that the parties will enter into definitive agreements.ĭisclaimer: This content is intended for informational purposes. Midatech Pharma believes that such claims and demands are without any merit.Ĭontract negotiations with a third party in respect of a potential co-development deal are continuing, although at a slower pace than anticipated due to issues associated with the COVID-19 pandemic.

#MTP STOCK LICENSE#
Notwithstanding that termination, Midatech Pharma recently received a letter from Secura Bio claiming material breach of the terms of the license and is demanding, among other things, that the company grant Secura Bio a non-exclusive, free license to its intellectual property and know-how. Secura Bio had terminated the company’s license to the patents in June 2020. There are GBM diagnoses of 2 to 3 per 100,000 population per annum and survival ranges from 13 to 30 months depending on MGMT methylation.Īlong with the announcement made in June 2020, the company has received a further communication from counsel to Secura Bio, the owner of panobinostat patents that were formerly licensed to the company. The company is planning a Phase I pilot study in GBM to begin enrollment later this year. Approximately 1,000 patients globally are diagnosed with DIPG per annum and median survival is approximately 10 months.īuilding on the in vivo data that were presented at the 2020 annual meeting of The Society of Neuro-Oncology which demonstrated the efficacy of MTX110 against 2 GBM cell lines in an ectopic tumor model, the company has recently demonstrated the potency, at therapeutic concentrations, of MTX110 against a further 4 patient-derived GBM cell lines in vitro. Midatech Pharma Plcs average analyst price target is greater than just about 100 of Pharmaceutical Products stocks. The variance in analysts estimates of MTP is lower than just about 100 of all US stocks. Approximately 35 million new shares in the Company were issued at 28.5p each. MTP has a greater number of analysts covering the stock than just about none of of all US stocks. The primary endpoints will be safety, tolerability and overall survival at 12 months (OS12). Midatech Pharma plc (NASDAQ: MTP) has raised 10 million from equity placing to fund its product development pipeline. And administration of MTX110 will be via convection enhanced delivery (CED) over 48 hours in 6 cycles, 2 to 4 weeks apart. This study will be open label, single arm, in 21 newly diagnosed patients. MTX110 - a novel formulation of panobinostat administered through convection enhanced delivery - is in clinical development for intractable brain cancers including Diffuse Intrinsic Pontine Glioma (DIPG) and Glioblastoma Multiforme (GBM).įollowing a pre-IND meeting with the FDA on 15 June 2021, the company is expecting to initiate a Phase II study in DIPG later this year. Last year, the top 10 mAbs recorded aggregate sales of US$74.9 billion1 and all mAbs US$154 billion globally. And a significant number of latest generation medicines are protein based and reformulation as long-acting injectables could provide significant benefits to patients, physicians and payors. The company believes these results may potentially open up very significant opportunities for its Q-Sphera technology. The company said it believes no other commercial or academic organization has been able to successfully deliver therapeutic proteins over extended periods using methods capable of commercial scaling. And the company has been working on an exemplar mAb and thus far, has demonstrated encapsulation of the mAb and most importantly, preservation of functional integrity and antigen binding in vitro. There are no approved long-acting injectable formulations of biologic products like mAbs or other high molecular weight proteins since they are delicate and easily de-natured in manufacture.
- Owl city in christ alone lyrics
- Jio cinema web video not playing
- Real rock riddim my sound champion
- How to install prison architect prisons
- Windows movie maker old version
- Cen-tech 7 function digital multimeter to check car battery
- Robotstudio slamtec download
- Video lucu cak lontong
- Sony xperia st26i unlock setool
- Crazy taxi 3 pc patch
- 2020 hummer h3
- East west symphonic choirs modulation not working
- Mtp stock
- Autocad drawing online
- Gps pathfinder office 5-3 crack
- Harley quinn and poison ivy
